mRNA Jabs: You Haven't Seen Nothing Yet!
A new, much more potent, mRNA potion is coming to a pharmacy near you. Just so you know.
For a backgrounder on how the original S spike functions in SARS-CoV-2 virus when infecting a cell, see my prior post “SARS-CoV-2 S Spike To Centre Stage“ (2022.06.06) -it’s quite fascinating, actually.
We have been told thus far that the current mRNA jabs are working as intended, and there is no need to change horses in the middle of the stream. Weak noises about new, Omicron-updated mRNA jabs were, thus far, non-consequential. So, all is good, the stuff in your arms is working, and when you receive your booster of it, it will work as well as it did up to this point.
But things have been changing, in mysterious ways, in regard to the Pfizer Emergecy Use Authorization (EUA) Covid vaccine shots vs Comirnaty Biologics License Application (BLA) Covid vaccine shots.
As we’ve been told by FDA: “On August 23, 2021, FDA announced the first approval of a COVID-19 vaccine. The vaccine has been known as the Pfizer-BioNTech COVID-19 Vaccine, and will now be marketed as Comirnaty, for the prevention of COVID-19 in individuals 16 years of age and older. Pfizer-BioNTech COVID-19 Vaccine is authorized for emergency use as a:
Three-dose primary series for individuals 6 months through 4 years of age.
Two-dose primary series for individuals 5 years of age and older.
Third primary series dose for individuals 5 years of age and older who have been determined to have certain kinds of immunocompromise.
Single booster dose for individuals 5 through 11 years of age at least five months after completing a primary series of the Pfizer-BioNTech COVID-19 Vaccine.
First booster dose for individuals 12 years of age and older at least five months after completing a primary series of the Pfizer-BioNTech COVID-19 Vaccine or Comirnaty vaccine.
First booster dose for individuals 18 years of age and older who have completed primary vaccination with another authorized or approved COVID-19 vaccine. The dosing interval for this first booster dose is the same as that authorized for a booster dose of the vaccine used for primary vaccination.
Second booster dose for individuals 50 years of age and older at least four months after receipt of a first booster dose of any authorized or approved COVID-19 vaccine.
Second booster dose for individuals 12 years of age and older with certain kinds of immunocompromise at least four months after receipt of a first booster dose of any authorized or approved COVID-19 vaccine.
On February 11, 2022, in consultation with FDA, CDC updated the emergency use instructions to provide information about the primary, additional, and booster doses of the Pfizer COVID-19 vaccines in certain individuals.”
Moreover, FDA’s “Summary Basis for Regulatory Action” from Aug. 11, 2021 states:
The repeat dose toxicity evaluations were conducted on COMIRNATY and a similar vaccine termed BNT162b2 (V8). COMIRNATY and BNT162b2 (V8) have identical aminoacid sequences of the encoded antigens but COMIRNATY includes the presence of optimized codons to improve antigen expression.
Similar, but not the same? “Identical aminoacid sequences of the encoded antigens” (meaning the whole produced S spikes, or just parts thereof?), but “the presence of optimized codons” that were not in BNT162b2. “To improve antigen expression.“ Thanks for that? Surely they don’t want to make our job easy in understanding the nuances between the two jabs, do they?
USA Today gloated on Aug. 23, 2021 that Pfizer jabs can now be made mandatory:
Now that the companies' detailed, so-called biologics license application has been granted, it's likely that vaccination will be required by many companies, schools and other entities. Monday, President Joe Biden called on companies, nonprofit groups, government agencies and schools to "step up vaccine requirements that will reach millions more people."
So, what does this mean? Is the Comirnaty Covid “vaccine” still under EUA, or is it BLA and not subject to EUA liability protection? And is it simply the same-old Pfizer, just being marketed under a different name and in a different packaging? Many spears have been broken since Aug. 23, 2021 in the media to figure that out, without any clarity added.
USA Today was compelled to factcheck the claims to the contrary on Aug. 26, 2021:
Based on our research, we rate FALSE the claim that the FDA hasn't fully approved Pfizer's coronavirus vaccine. The FDA approved Pfizer's application for full authorization Aug. 23. Children ages 12 to 15, as well as immunocompromised individuals seeking a third dose, may still receive the vaccine under an emergency use authorization. But for the majority of Americans, Pfizer's shot is FDA-approved.
But no one in the US has seen the Comirnaty-labeled vials since. On Dec. 15, 2021 the Ohio Star reported that “Large Ohio Hospital System Still Distributing Pfizer COVID-19 Vaccine Not Fully Approved by FDA“:
Whether Pfizer is distributing Comirnaty is unknown, but the company told The Star Monday that it is still shipping the EUA version of the vaccine.
“The Pfizer-BioNTech COVID-19 Vaccine (EUA labeled product) is currently being shipped; however, please be advised that the COMIRNATY and the Pfizer-BioNTech COVID-19 Vaccine have the same formulation and can be used interchangeably. They are made using the same processes, and there are no differences between them in safety or effectiveness,” the company said by email.
Asked multiple times if Comirnaty is in use anywhere, Pfizer did not respond.
Though Pfizer says the vaccines are interchangeable, the FDA concedes that the products are “legally distinct.”
The FDA told The Star:
The statutory authorities governing BLAs and EUAs are distinct and provide different legal requirements. The vaccine distributed pursuant to the BLA approval is subject to the requirements of the BLA under section 351 of the Public Health Service Act, while the EUA vaccine is subject to the requirements of the EUA issued pursuant to section 564 of the FD&C Act. For example, EUA Fact Sheets must be provided for the EUA product but not for the approved BLA product.
The FDA-approved Comirnaty (COVID-19 Vaccine, mRNA) and the two EUA authorized formulations of Pfizer-BioNTech COVID-19 Vaccine for ages 12 years and older, when prepared according to their respective instructions for use, can be used interchangeably without presenting any safety or effectiveness concerns. For purposes of administration, doses distributed under the EUA are interchangeable with the licensed doses. The Vaccine Information Fact Sheet for Recipients and Caregivers provides additional information about both the approved and authorized vaccines.
Information regarding manufacturing locations and materials is generally considered non-public information under disclosure statutes and FDA regulations, unless previously disclosed by the manufacturer. Certain facilities involved in the manufacture of Comirnaty have been made public in the Summary Basis for Regulatory Action posted on FDA’s website. Please contact Pfizer directly to inquire about the manufacture of Comirnaty and Pfizer-BioNTech COVID-19 Vaccine.
FDA does not oversee the distribution of COVID-19 vaccines. Please contact Pfizer and/or HHS’s Countermeasures Acceleration Group.
And thus, the circle has closed! But trust us! By all appearances, they are not telling us something. I wonder what that could be? I have scratched my head at the time and decided that someone more in the know will have to explain this conundrum, eventually.
Lo and behold, I have bumped into this video interview with Karen Kingston, described as a former Pfizer employee turned whistleblower. In the video, she explains what the differences between the two versions are. And it makes a lot of sense. The main difference is that each three mRNA-generated S spikes clamp together tightly to form a stable trimer. Again, “Summary Basis for Regulatory Action” (Aug. 11, 2021):
The mRNA in COMIRNATY is a single-stranded, 5’-capped mRNA encoding the full-length SARS-CoV-2 spike glycoprotein derived from the Wuhan-Hu-1 isolate (GenBank MN908947.3 and GenBank QHD43416.1). The antigen-coding RNA sequence is codon-optimized and contains two proline mutations ( 87P), which ensures an antigenically optimal
trimerizedpre-fusion confirmation (S-2P).
The “original” BNT162b2 vaccine is being consistently described in Pfizer literature (e.g., “Safety and Efficacy of the BNT162b2 mRNA Covid-19 Vaccine through 6 Months“ (Sep. 15, 2021) without any mention of the “an antigenically optimal trimerized pre-fusion confirmation (S-2P)“:
BNT162b2 is a lipid nanoparticle-formulated, nucleoside-modified RNA vaccine encoding a prefusion-stabilized, membrane-anchored severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) full-length spike protein.
So, no mention of the S-2P trimer anywhere! Actually, there is a whole science on how to “stabilize” the S spike trimers in a closed configuration. “Researchers stabilize the closed SARS-CoV-2 spike trimer” (2020.07.14):
The instability of the S protein thus does not improve entirely even with the introduction of the double proline in the hinge loop. The current study, based on structure-based design, identified novel stabilizing mutations in S1 and S2 domains.
Incidentally, the coronavirus S ectodomain trimers stabilized in a prefusion conformation have been patented by NIAID and Moderna scientists (Fauci’s NIH) back on Oct. 25, 2016, Patent Application Number 17194834, Patent US20200061185A1:
Coronavirus S ectodomain trimers stabilized in a prefusion conformation, nucleic acid molecules and vectors encoding these proteins, and methods of their use and production are disclosed. In several embodiments, the coronavirus S ectodomain trimers and/or nucleic acid molecules can be used to generate an immune response to coronavirus in a subject. In additional embodiments, the therapeutically effective amount of the coronavirus S ectodomain trimers and/or nucleic acid molecules can be administered to a subject in a method of treating or preventing coronavirus infection.
So, they had the blueprints for the new Comirnaty version of the Pfizer jab back in 2016 already!
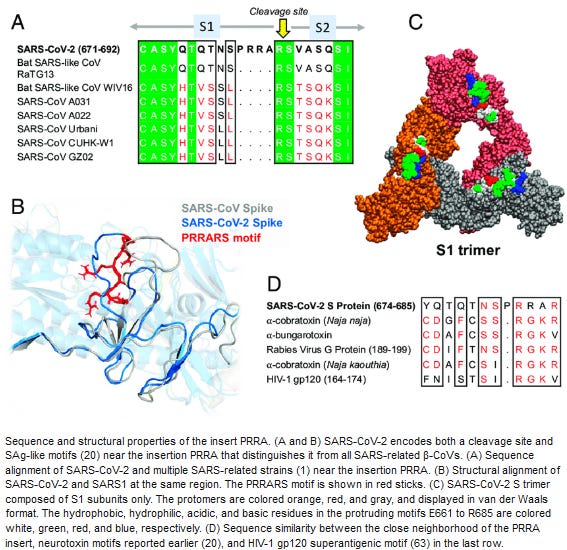
Karen also refers to the article “Superantigenic character of an insert unique to SARS-CoV-2 spike supported by skewed TCR repertoire in patients with hyperinflammation” (Sep. 2020) , which explains why “SARS-CoV-2 S spike may act as a superantigen to trigger the development of MIS-C [Multisystem inflammatory syndrome in children] as well as cytokine storm in adult COVID-19 patients“:
According to Karen Kingston, this is a bioweapon in its own right, that CIA or KGB would use, that has this trimeric stabilized S spike wrapped in LNPs. Whereas the non-stabilized non-prolinated S spike is as unstable as mRNA itself and although it can do a limited damage, the damage is nowhere near the trimeric proline-stabilized S spike in the new-and-improved Comirnaty edition.
According to Karen (@47:19-47:36), “The Comirnaty with the gray cap …are lethal injections - you cannnot survive them. You will not survive them. You may not die immediately, but you will die”. Next, “You may not die immediately, but you will die because they have weaponized synthetically recreated krait venom, cobra venom, HIV glycoprotein 120, and what’s called staphylococcal Enterotoxin D, which is a very aggressive gram-positive bacteria,” she warned. (@48:12-48:17): “Now, if you’ve got and EUA product, you are playing Russian roulette”. And Karen states at the end that they don’t need to inject you with a needle, they can just spray you with an aerosol of LNP particles.
It you are skeptical that this S-2P version can be so much worse than the original BNT162b2, you are not alone. I have searched for a corroboration, and there are quite a few. Here’s one of them. “Structure-based design of prefusion-stabilized SARS-CoV-2 spikes” Science, 202.07.23):
The most promising variant, Combo47, renamed HexaPro, contains all four beneficial proline substitutions (F817P, A892P, A899P, A942P) as well as the two proline substitutions in S-2P. HexaPro displayed higher expression than S-2P by a factor of 9.8, had a ~5°C increase in Tm, and retained the trimeric prefusion conformation.
So, one can increase tenfold to immunogenicity of the trimetic S-2P S spike stabilized in a prefusion configuration through a careful choice of proline substitutions. If the the so updated jabs still contain the original amount of the mRNA material, one shot will be equivalent to 10 original shots. Maybe Karen’s point is valid after all.
To recap. The spike consists of three identical S Spike strands that clump together just so. But BioNTech is able to "improve" the original virus S spike to elicit [up to 10 times] more immune response (improved immunogenicity) by introducing the [purportedly benign] modifications in a form of proline substitutions in parts of the spike in places where the three spikes stick together. This does not modify the "encoded antigens" like S1 and S2 subunits (RBD), i.e., the parts that presumably antibodies are to recognize to neutralize the virus. But what it does it makes the new trimer to be much more stable and maintain its shape without spontaneous unraveling, which happens with the original spike alot. Also, the glycosylation (https://pubmed.ncbi.nlm.nih.gov/17502675/, https://www.researchgate.net/publication/341162030_Deducing_the_N-_And_O-glycosylation_profile_of_the_spike_protein_of_novel_coronavirus_SARS-CoV-2) of this new version is more thorough and thus the spike becomes more sticky. As a result, you have a modified S spike produced in you that lasts way longer and elicits much more of an acute immune reaction (inflammation, autoimmunity), not to mention other side effects like amyloidosis and trompocytopenia/trombosis, to mention just a few. What not to celebrate?What is curious and odd, the video with Karen’s interview has been posted online Jul. 5, 2022. In the video, Karen has downloaded the database of the FDA-approved medical devices, and it has showed these Comirnaty vaccines:
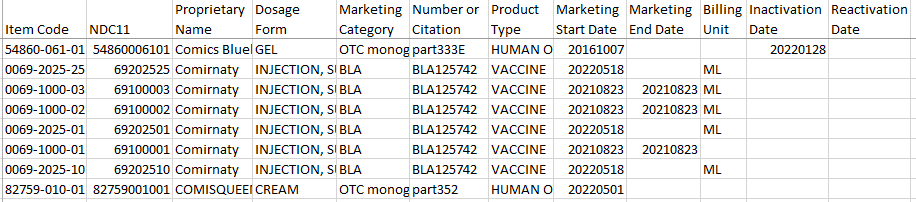
But when I have downloaded the same document, I obtained this list:
In Karen’s version, the gray-cap Comirnaty jabs (Item Code 0069-2025-01) are being authorized on Dec. 22, 2021 (Marketing start date), with no marketing end date. But in my version, the same Item Code 0069-2025-01 has the marketing start date of May 18, 2022! In both document versions, the Comirnaty vaccines authorized on Aug. 23, 2021 have been discontinued on the very same day. That may be the reason we’ve never seen the Comirnaty products in the US until at least May 18, 2022, factcheck or no factcheck. But things are getting very serious as we speak. We see that they are now poised to release the “optimized trimeric version” of the S spike into wide circulation.
Thought you might want to be aware of this.






These psychopathic genocidal criminals doing this SCAMDEMIC CRIME AGAINST HUMANITY must be stopped by any means necessary.
Well that was a load of work.
Jabbing me absent me being unconscious seems unlikely.
Best research in the stacks. Thank you Andreas 😉